|
|

On May 24th, Nadia Guarini publicly presented her PhD work and successfully received her diploma. Her research dealt with the molecular relationships between membrane transporters and TORC1, a kinase complex playing a pivotal role in control of cell growth. More specifically, she focused on initial TORC1 activation upon nutrient uptake into yeast cells and the important role in this process of the most abundant cell surface protein, namely the H+-pump encoded by the PMA1 gene. The signal of this activation is the influx of H+ coupled to active nutrient uptake via permeases and sensed by the Pma1 H+ pump. Nadia has solved the long-sought question of what are the protein kinases and phosphatases mediating Pma1 phosphoregulation and she also uncovered an interesting negative feedback control of Pma1 by activated TORC1. Congratulations to Nadia for her scientific achievements and thanks to her for the excellent time spent with her in the lab !
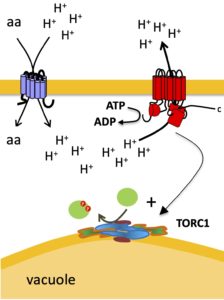 The yeast Pma1 H+-ATPase energizes the plasma membrane and is crucial for cell viability. This H+ pump is also involved in initial TORC1 activation upon H+-coupled active uptake of nutrients into starved cells. In this article entitled “Phosphoregulation of the yeast Pma1 H+-ATPase autoinhibitory domain involves the Ptk1/2 kinases and the Glc7 PP1 phosphatase and is under TORC1 control“, we identify the largely redundant Ptk1 and Ptk2 kinases and the Glc7 PP1 phosphatase as the key actors of the phosphoregulation of Pma1. We also unravel that this phosphocontrol, when defective, negatively impacts Pma1-mediated TORC1 regulation, and that TORC1 feedback inhibits Pma1 phosphorylation. This work has been largely carried out by Nadia Guarini thanks to initial observations by Elie Saliba. The yeast Pma1 H+-ATPase energizes the plasma membrane and is crucial for cell viability. This H+ pump is also involved in initial TORC1 activation upon H+-coupled active uptake of nutrients into starved cells. In this article entitled “Phosphoregulation of the yeast Pma1 H+-ATPase autoinhibitory domain involves the Ptk1/2 kinases and the Glc7 PP1 phosphatase and is under TORC1 control“, we identify the largely redundant Ptk1 and Ptk2 kinases and the Glc7 PP1 phosphatase as the key actors of the phosphoregulation of Pma1. We also unravel that this phosphocontrol, when defective, negatively impacts Pma1-mediated TORC1 regulation, and that TORC1 feedback inhibits Pma1 phosphorylation. This work has been largely carried out by Nadia Guarini thanks to initial observations by Elie Saliba.

Christopher Dereppe and Bruno André participated to the last “TOR de France” meeting organized at Nice on 5-6 October. This international meeting gathers every two years, in a nice place in France, scientists studying the “Target Of Rapamycin” (TOR) kinase complex involved in control of cell growth and associated with several diseases including cancers. The meeting was of high interest and offered an opportunity to meet again many colleagues interested in TOR.

Luis Sousa, Christopher Dereppe and Bruno André participated to the 38th international “Small Meeting on Yeast Transport and Energetics (SMYTE)” organized last September in Blankenberge (Belgium) by Pr. Patrick Van Dijck (KUL, Belgium). The Smyte annually gathers scientists from all countries who are interested in diverse aspects yeast membrane transporters: structure-function relationships, regulation, and signaling properties. Luis presented a poster about his work on amino-acid excretion by yeast cells and Christopher and Bruno gave presentations about the control of TORC1 in yeast.
We are delighted to announce that the laboratory has been awarded new funding to study the relationship between cell aging, the lysosome and TORC1 in yeast. This project is being carried out in collaboration with Prof. Cédric Delporte at ULB’s Faculty of Pharmacy. The initial team made of Dimitrios Zisimopoulos (postdoc) and Charlotte Felten (technician) now includes Chloé Troch (PhD student) and Alexandre Deschamps (pregraduate student). Chloé has completed her Master last year in the laboratory and has been awarded an FNRS “Aspirant” grant. She was also awarded the prize for the best Master’s student – double congratulations to her!
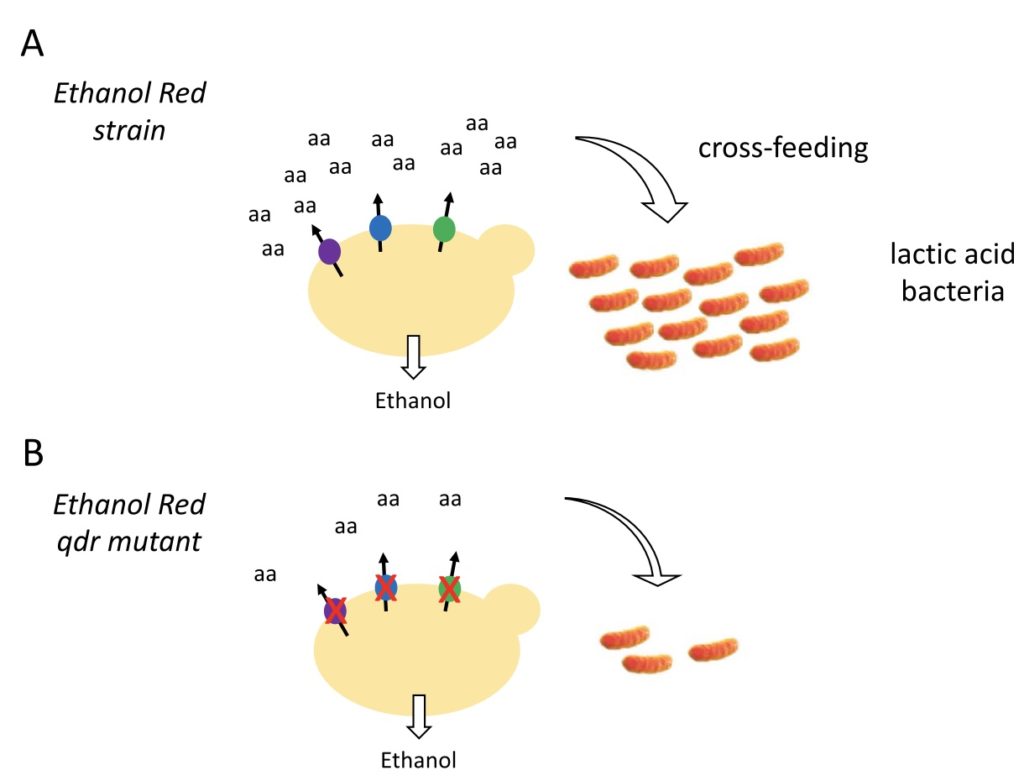
We are pleased to announce the publication in Scientific Reports of an article entitled “Deletion of QDR genes in a bioethanol‐producing yeast strain reduces propagation of contaminating lactic acid bacteria“. This study reports that the yeast Ethanol Red strain commonly used to produce bioethanol excretes amino acids via Qdr membrane transporters. This excretion favors propagation of co-cultivated lactic acid bacteria, which cannot grow without an external supply of amino acids. A derivative yeast mutant in which QDR genes have been deleted via CRISPR-Cas9 displays markedly reduced cross-feeding of these bacteria without reduction of ethanol production. Given that bacterial contaminations in yeast fermentation tanks are a recurring problem for the bioethanol production industry, this finding opens the perspective of using yeast strains with mutations in QDR genes to reduce the risk of such contaminations. This study, which has been mainly conducted by Dr. George Kapetanakis during his PhD work, is the result of a fruitful collaboration with Dr. Isabelle Georis (Labiris), Dr. LaurenceVan Nedervelde (Labiris), and the “Syngulon” startup company.

On September 30th, George Kapetanakis (on the right in the picture) publicly presented his PhD work and successfully received his diploma. His research dealt with the poorly known process of amino-acid excretion by yeast cells. He characterized two novel amino-acid exporters and showed that these proteins importantly contribute to cross-feeding by yeast of co-cultivated lactic acid bacteria which cannot grow without an external supply of amino acids. G Kapetanakis also showed that the same proteins mediate excretion of amino acids and cross-feeding of lactic acid bacteria by the Ethanol Red yeast strain used in industrial bioethanol production. This observation is important since contamination of fermenters by lactic acid bacteria is a major issue in bioethanol production. We thus proposed in a novel patent of ULB that the utilization of yeast mutants lacking specific amino-acid exporters reduce the risk of contaminations of bioethanol production tanks by lactic acid bacteria. George Kapetanakis has now been hired as Research and Innovation Scientist in the young Paleo startup (see previous post). Congratulations and thanks to George for his scientific achievements and their potential industrial applications, and good luck with the new job in Paleo!

The startup company Paleo exploits yeast to produce GMO-free, heme proteins of beef and other animals for plant-based meat alternatives, with the ultimate goal of making food production more ethical and sustainable. Paleo recently moved into the lab space and offices next to our lab in the IBMM (Institute of Molecular Biology and Medicine). We established excellent contacts with the Paleo team members and we look forward to collaborate with them. Welcome to Paleo and great success in the future!

Nadia Guarini, Luis Sousa, Christopher Dereppe and Bruno André participated to the Interuniversity PhD student day organized in Namur last September 9th. As most other PhD students who attended this event, Nadia, Luis and Christopher presented a flash talk and a poster. This PhD student day, which was cancelled the last two previous years, was a big success.

Nadia Guarini attended the Yeast Genetics meeting 2022 organized at University of California in Los Angeles (August 17–21). She presented a “flash-talk” and a poster about her work on the role of the yeast H+-ATPase (Pma1) in control of TORC1 in yeast. This conference, the first edition of which was organized 40 years ago, is the “premier meeting for students, postdoctoral scholars, research staff, and principal investigators studying various aspects of eukaryotic biology in yeast, a major experimental model for understanding human cell biology and mechanisms of human disease“.
|
|









